  You learned about the wave properties of matter, and how to describe each individual electron in terms of its four quantum numbers. 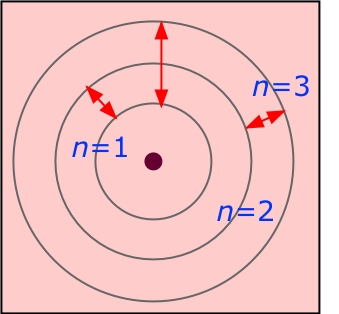
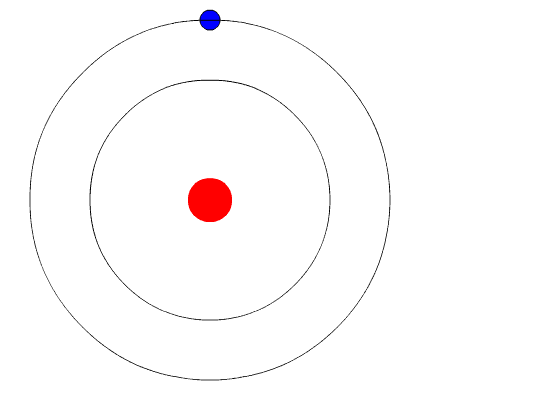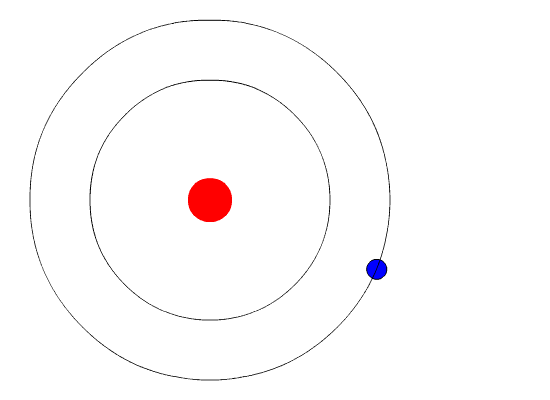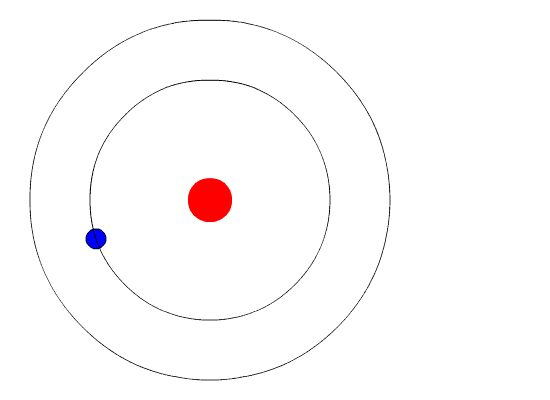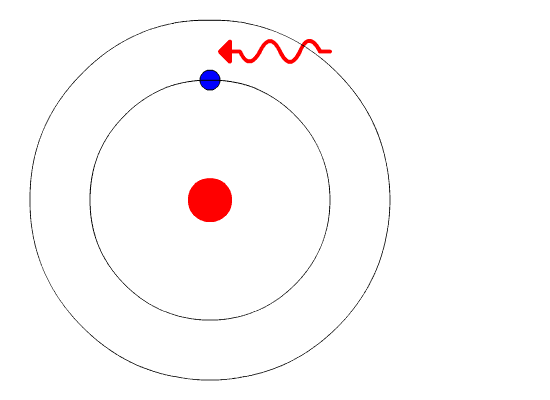
In this chapter, you learned about the electronic structure of the atom in terms of the older Bohr model and the newer quantum mechanical model. It is addressed in the Bohr- Sommerfield model, and is the result of a quantum mechanical interaction, known as spin-orbit coupling, further discussion of which is not necessary for this volume. This is termed fine structure, and is not predictable from the Bohr model of the atom. Īs a final note, closer inspection of the emission lines from Na shows that most emission lines are not, in fact, single lines, but are closely spaced doublets or triplets - for example, the strong yellow line discussed above at 589.3 nm is composed of two separate lines at 589.0 and 589.6 nm. For the heavier elements in the periodic table, the absorption and emission spectra can be extremely complicated - manganese and iron, for example, have about 4600 lines in the visible and UV region of the spectrum. Taken together, the Bohr model of quantized electron orbitals, the selection rules, and the relationship between wavelength and energy derived from particle-wave duality are sufficient to explain the major features of the emission spectra of all elements. Thus the 3s -> 3p transition is allowed, although the 3s -> 4s is forbidden, since in this case A/ = 0 and is forbidden. However, in the sodium atom, An = 0 is also allowed. These energy levels differ in detail between atoms of. The Bohr model shows that electrons can only occupy orbitals whose energy is fixed (quantized), and that each atom is characterized by a particular set of energy levels. Īppendix 1 also shows how the periodic table of the elements (Appendix 5) can be built up from the known rules for filling up the various electron energy levels. All of these exactly known quantities-position, distance from nucleus, energy, and velocity-can t, according to the Heisenberg uncertainty principle, be known with great precision simultaneously. And finally, the velocity of the electron in its orbit is exactly known. 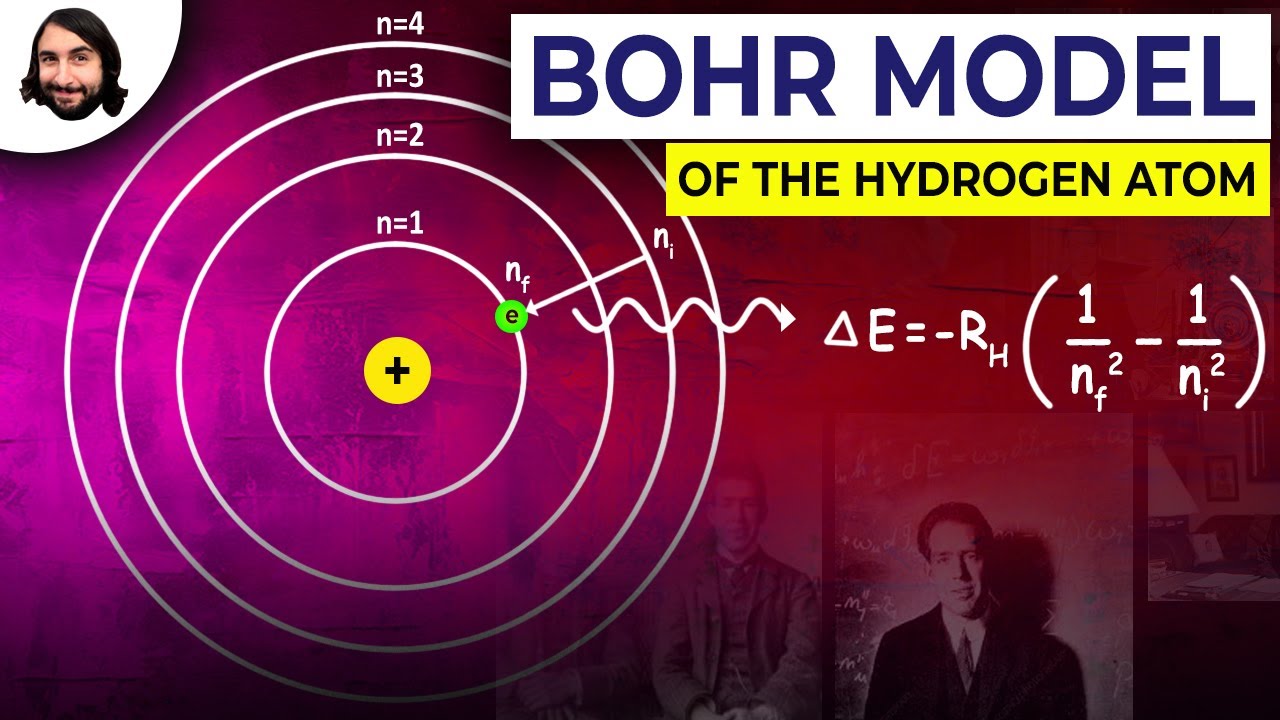
The distance of the electron from the nucleus also is exactly known, as is its energy. It implies that the position of the electron is exactly known at any time in the future, once that position is known at the present. The Bohr model is a determinant model of an atom. The quantum number n is essentially equivalent to the n that was assumed in the Bohr model of hydrogen. Note that from the solution of a problem involving three dimensions, three quantum numbers result, unlike the Bohr approach, which specified only one. When solved for m, this balancing equation is. This assumption is the basis of the Bohr model for the hydrogen-like atom. The biggest problem with the Bohr atom, however, lay in its lack of a. Although scientists could calculate the emission spectrum of hydrogen using the Bohr model, the model could not account for the spectra of heavier atoms. The Bohr atom went a long way toward explaining the nature of atoms, but there were problems. For example, conceptual understanding of nature of different types of chemical bonding, atomic theory in terms of the Democritus particle model and the Bohr model, and. Although these visualization skills are very important in chemistry, it is evident that they are not the only ones needed in school chemistry (Mathewson, 1999). In chemistry, perhaps because of the significance in visualizing molecular strac-ture, there has been a focus on how students perceive three-dimensional objects from a two- dimensional representation and how students mentally manipulate rotated, reflected and inverted objects (Stieff, 2007 Tuckey Selvaratnam, 1993). Īccording to the Bohr model, the radius of a circular orbit is given by the equation. įrequency-wavelength Energy-frequency Bohr model. Use the Bohr model to identify lines in the hydrogen spectrum. In particular, according to quantum mechanics. The quantum mechanical atom differs from the Bohr model in several ways. īefore proceeding with the Bohr model, let us make three points. It is logical to assume that it is the outer edges of the atomic spheres, and thus the electrons, that interact when there is a close approach between the atoms, ultimately resulting in chemical combination. As stated previously, with the protons and neutrons in the nucleus deep inside the atom, it is logical to think that the electrons play the most important role as far as contact with other atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
